

responses and
tumor control




Make every CLL/MCL patient eligible for safe and effective CAR-T cell therapy. With our first-in-class 5th generation Tandem CAR, we are going to redefine current lymphoma treatment. This will be accomplished by targeting a combination of novel tumor-specific antigens and introducing a novel signaling domain. As a result, our TanCAR confers superior CAR T-cell eradication of CLL cells, sparing healthy cells and limiting cell-therapy related negative effects and consequences.
Our Solution
Immunofusion’s 5th generation TanCAR encompasses two distant technologies – novel tandem targeting module – TanCLL and an engineered signaling module – ILX-cel, that are solving the major issues of existing CAR-T therapies
ILX-cel technology
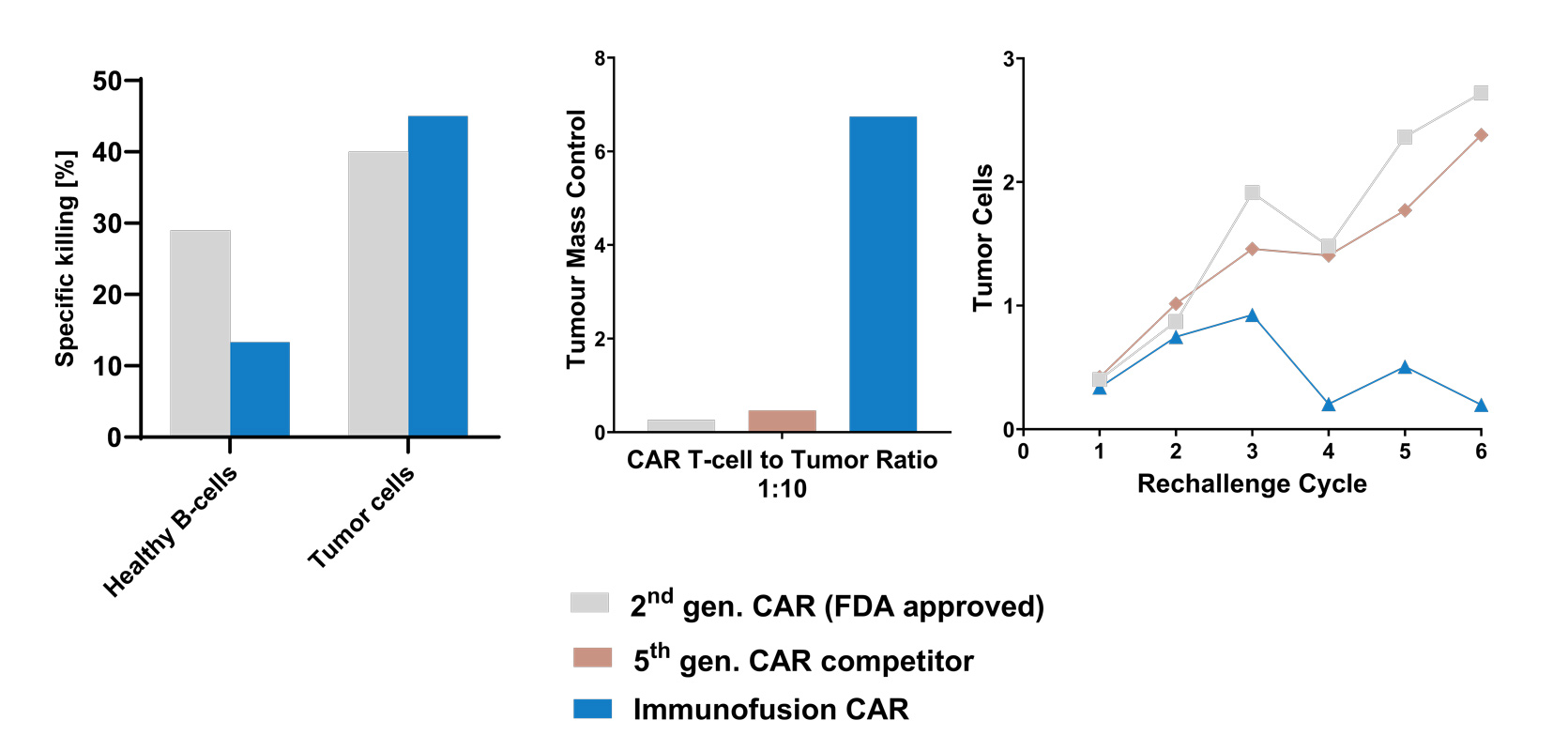
Long-lasting responses and tumor control
With our 5th generation CAR-T therapy, patients can expect durable responses, prolonged remission periods and cure. Our innovative design promotes an enhanced T-cell expansion/persistence and killing capacity, reducing the likelihood of disease relapse and offering patients long-term peace of mind
Accessibility
Our novel signaling domain enhances the expansion and functionality of even extremely exhausted T cells, potentially increasing the number of patients eligible for life-saving CAR T-cell therapy and enabling the use of lower dosages in clinical applications.
TanCLL technology
Enhanced Efficiency
By leveraging state-of-the-art engineering, our CAR-T therapy combines very tumor-specific antigen targeting and CAR-T armoring for high potency against high tumor burden (adverse factor for all 2 generation CAR-T cells), minimization harm to healthy tissues and at the same time lowering the possibility of antigen-negative relapses (drawback of all 2 generation CAR-T cells) posing a bold aim of patients cure.
Tailored Treatment
Every patient will obtain the most specific CAR-T cell product matching their lymphoma antigen repertoire ensuring unprecedented efficacy and response rates
Superior Safety Profile
We’ve prioritized safety every step of the way. Our advanced and tumor-specific design mitigates the risk of adverse events commonly associated with traditional CAR-T therapies, such as B-cell aplasia and subsequent infections, offering patients a smoother treatment journey with reduced chances of complications
Continual Innovation
We’re committed to pushing the boundaries of CAR-T therapy, far beyond our TanCAR main indication. Now we are ready to expand our technology to the autoimmune disease field by precisely targeting only specific and pathogenic B-cells instead of depletion of all B-cells
Unmet medical need in CLL\MCL treatment
CAR-T manufactoring issues, up to (%)
CAR-T cell manufacturing failure
0%
Low quality starting material and a final product
0%
CAR-T treatment failures, up to (%)
Low expansion and exhaustion (IC)
0%
High tumor burden
0%
Infections as a leading cause of non relapse mortality
0%
Antigen escape
0%
News

Nov 2024 – Immunofusion has been selected as a semifinalist for @EITHealth’s Catapult 2024
We’re eagerly preparing for our upcoming training sessions with industry experts, where we’ll refine our pitches to compete for a spot in the finals in front of a panel of investors.

Nov 2024 – Immunofusion visited Biopark Charleroi Brussels South (ULB)
Immunofusion visited Biopark Charleroi Brussels South (ULB) to explore establishing a wet lab in the heart of the Wallonia region.

March 2023 – Immunofusion is a finalist of the BIO EUROPE-2023 Startup Spotlight
Immunofusion is thrilled to be selected as one of eight biotech startups across Europe with the opportunity to engage with top-tier investors and key players in the biotech industry

July 2024 – Immunofusion drafted the ILX-cel domain patent application
Immunofusion is thrilled to be selected as one of eight biotech startups across Europe with the opportunity to engage with top-tier investors and key players in the biotech industry
Leadership
Where Passion and Professionalism Meet Unmet Medical Needs
Make every CLL/MCL patient eligible for safe and effective CAR-T cell therapy. With our first-in-class 5th generation Tandem CAR, we are going to redefine current lymphoma treatment. This will be accomplished by targeting a combination of novel tumor-specific antigens and introducing a novel signaling domain. As a result, our TanCAR confers superior CAR T-cell eradication of CLL cells, sparing healthy cells and limiting cell-therapy related negative effects and consequences.

Dmitri Dormeshkin, PhD
10 year antibody engineering for CART, IVD and R&D.

Mikalai Katsin, MD
Head of the hematology/oncology department 5 years, PI/SI in multiple phase I/II/III trials.

Alex Migas, PhD
Cellular immunotherapy (CAR-T, NK-CAR) local manufacturing for clinical applications.

Alexander Meleshko, PhD
Head of Preclinical Models and Cancer Vaccines Unit
Are you interested in our solution?
Do you share our vision or want to support our mission?
Reach out to start a conversation.
